In her lab at the Phil and Penny Knight Campus for Accelerating Scientific Impact, computational biochemist Parisa Hosseinzadeh is using computer modeling to design synthetic peptides as potential drugs to treat challenging diseases.
Hundreds of synthetic peptides are either in use or in clinical trials, but producing them is time-consuming and costly. Vaccines against COVID-19 have peptides that target the spike protein of the virus, tricking it and thwarting infection.
Peptides are tiny molecules that contain two or more amino acids, the building blocks of proteins. They emerged as a therapeutic in 1922, when a natural, hormone-produced version in pigs was used to regulate insulin in a child with diabetes. The first lab-produced peptide came in 1953 and is commonly used to induce labor.
Hosseinzadeh, an assistant professor, is deploying computational methods to help drug designers move more swiftly in their screening of possible peptides that will bind precisely to targets so they initiate a desired response without causing unintended consequences.
“Researchers have been generating huge libraries of random peptides and then screening them to see if they bind to a target or not. It is a random, trial-and-error process,” she said. “The problem with this method is that the overall space in which you can screen for candidate peptides is vast, like 10 to the 30th (power). In a best-case scenario, we can screen 10 to the 14th. It is impossible to screen everything.”
Her research at the Knight Campus aims to narrow that testing space of more than a hundred trillion possibilities to a smaller and more manageable pool of possible candidates. It builds on work she began before arriving at the UO last September.
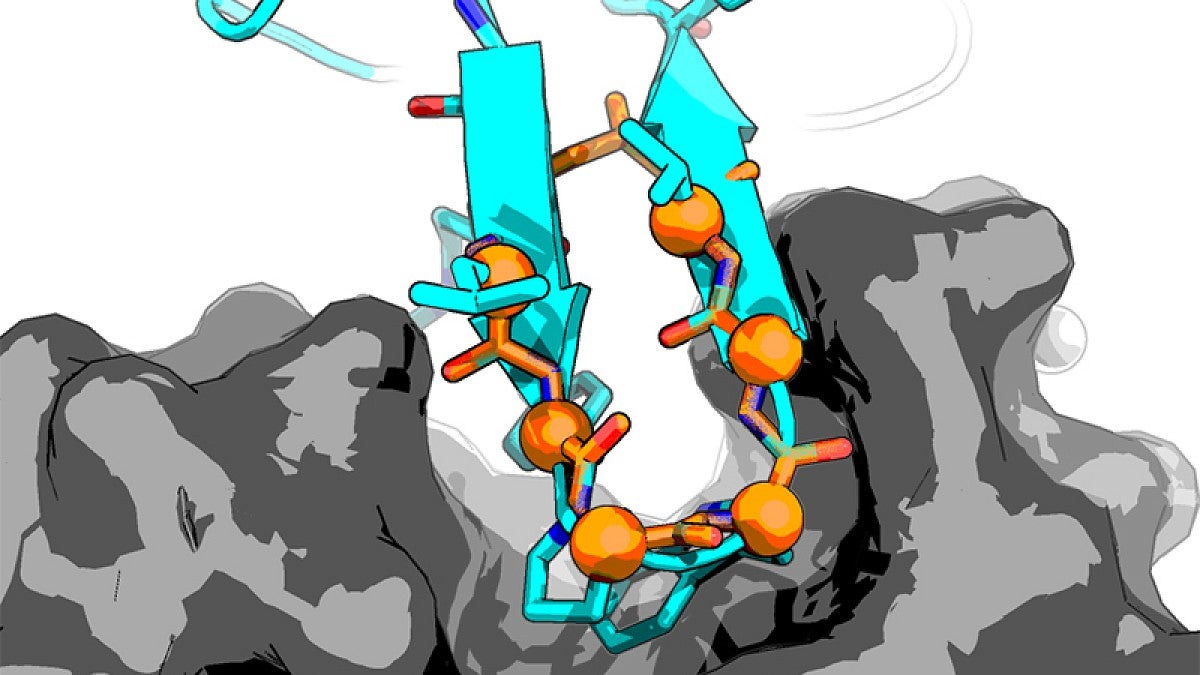
Hosseinzadeh has emerged as a leader in her field. While a postdoctoral researcher at the University of Washington, she led a study published in 2017 in the journal Science that led to highly structured, rigid peptides with high accuracy. That work was done with UW colleagues, Howard Hughes Medical Institute investigators and scientists at the Pacific Northwest National Laboratory.
“Accurate design of structured peptides in this scale had never been done before, and many people thought it couldn’t be done,” she said. “If you have something that is rigid you can better predict its behavior with computational analyses. This rigidity allows for tighter binding.”
This year has brought two new published studies from work done prior to her arrival in Eugene.
In a paper published online June 7 in Nature Communications, Hosseinzadeh, in her postdoctoral role at the UW, and colleagues from the UW, University of Pennsylvania and Stanford University unveiled a proof-of-concept for a computational approach that reduces the screening time for peptides that will bind.
Her team detailed how their approach used computational modeling to screen hundreds of thousands of peptides, generating 50 candidate peptides for testing. The computations considered all possible combinations of the human body’s 20 amino acids and associated compounds.
Each tested peptide was designed with a non-protein-generating amino acid as an anchor to provide a weak initial binding around which peptides can be designed to enhance the binding.
“Using this method,” Hosseinzadeh said, “we obtained peptides that can inhibit a class of enzymes with low nanomolar affinity without any downstream optimization."
Low affinity at nanometer scale is important in drug design; it means that a peptide can bind only at a targeted site without affecting other proteins that lead to undesired effects.
“Our method involves fewer experiments and is faster,” she said. “We can weed out candidate peptides in the experimental libraries that will never bind. At this stage, our work is about creating a platform that scientists can use to generalize to meet their specific needs.”
In a paper published March 25 in ACS Catalysis, a journal of the American Chemical Society, Hosseinzadeh and colleagues addressed the applications of peptide design, particularly in probing the mechanism of enzymatic reactions. Their computational modeling and experimental techniques enabled an investigation of the activity of multiple conformations of a peptide catalyst in isolation.
“We determined that the dynamic movement of the lead catalyst plays a crucial role in achieving a site-specific reaction,” Hosseinzadeh said. “This approach may also serve as a valuable method for investigating the mechanism of other peptide-catalyzed transformations."
Researchers in Hosseinzadeh’s Knight Campus lab are focusing on the fundamentals of synthetic peptide design. They are using computational modeling and analyses to study peptide behavior. She also is seeking to develop peptide- and protein-based biosensors for disease diagnostics.
Peptide-based vaccines are in play or under development by numerous researchers to treat diseases such as influenza, cancers, hepatitis C, HIV and brain disorders.
Synthetic peptides, she said, hold promise as a treatment for disease targets currently out of range of current drugs.
“At this point, my main focus is developing robust and accurate computational methods, but, as this work develops, I look forward to working with anyone who is pursuing specific targets,” Hosseinzadeh said.
She is already collaborating with Knight Campus colleagues who are interested in specific binding capabilities.
“The collaboration with colleagues at the Knight Campus is a huge motivation for me to move this research forward,” she said. “It’s nice to be around people who are more applied in their focus. I can talk with them and ask questions about next steps I might take. It often leads to questions that bring about new challenges that I have not thought about before.”
—By Jim Barlow, University Communications