Imagine a drug that could temporarily reenergize plasticity in the brain to treat autism or schizophrenia, or even help an adult’s aging brain pick up a new language or learn to play a musical instrument.
Such are the potential, down-the-road medical implications of a discovery made by a postdoctoral researcher in the Institute of Neuroscience lab of Chris Doe, a Howard Hughes Medical Institute investigator and professor in the UO’s Department of Biology.
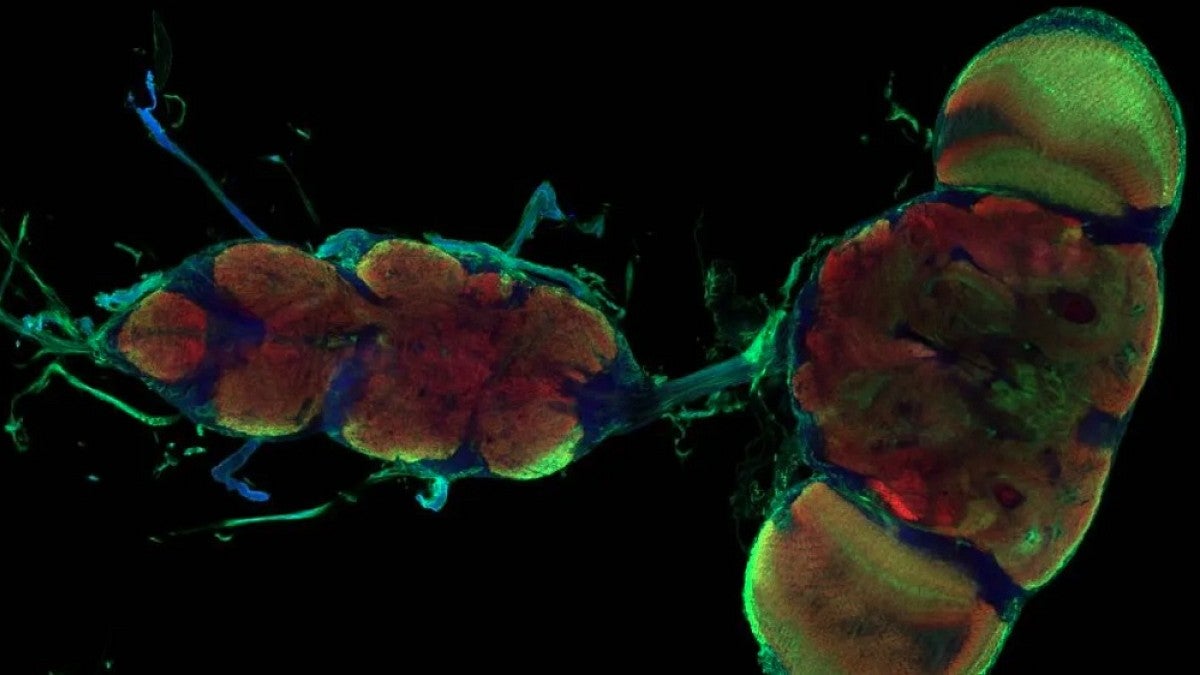
In a basic science study detailed April 7 in the journal Nature, Sarah Ackerman identified nonelectrical cells that transition the brain from a highly plastic state, open to rapid learning, into a less moldable, mature state in the developing central nervous system of fruit flies.
The cells, known as astrocytes for their star-like shapes, and associated genes eventually could become therapeutic targets, Ackerman said.
“All of the cell types and signaling pathways I looked at are present in humans,” she said. “Two of the genes that I identified are susceptibility genes linked to neurodevelopmental disorders including autism and schizophrenia.”
The failure to close so-called critical periods of brain plasticity in development also is associated with epilepsy, she said.
“In childhood, you can rapidly learn new tasks, remember things and learn new languages, all of which becomes harder as we age,” Ackerman said. “The reason for that is that the circuits in the young brain are really plastic; they can change in response to experience and activity. I am interested in the mechanisms that regulate that shift from that plastic and dynamic state to the more stable adult state.”
Astrocytes are glial cells found in large numbers in the central nervous system. They have diverse roles that are based on where in the brain and spinal cord they are active. They are, Ackerman said, “the guardians of synapses in terms of assuring proper functioning in both their formation and later performance.”
In the research, Ackerman focused on the motor circuitry of Drosophila fly larvae over specific points in development. These invertebrates are standard research models that allow rapid genetic exploration of molecular mechanisms using human-related genes.
Ackerman used light-based optogenetics to selectively turn motor neurons off and on. The neurons, she said, exhibited striking changes to their shape and connections — their plasticity — in response to the manipulations.
Curiously, Ackerman’s team saw astrocytes pouring into the nervous system, extending fine projections and enveloping neuronal connections just as the circuitry switches from a plastic to stable state. Ackerman then screened for candidate genes associated with astrocytes to determine which molecular pathways direct the window to close and shut down motor plasticity.
That work pointed directly at neuroligin, a protein on astrocyte projections, that binds to neurexin, a receptor protein on dendrites from developing neurons. Eliminating that genetic pathway extended plasticity, while precocious expression of these proteins closed plasticity too early in development. Both proteins are present in the human nervous system.
Changes in the timing of plasticity also were found to later impact behavior. A brief extension of plasticity resulted in abnormal crawling of the fly larvae several days later. Extending periods of plasticity in human development, Ackerman said, are tied to neurodevelopmental disorders.
A tragic human example of how vital this critical period is, Doe said, may be the case of abandoned Romanian children found in an orphanage in the 1980s. Hundreds of babies had been neglected except when they were fed or washed, according to news reports.
The neglect would have occurred during that key period of plasticity when experiences and learning mold the brain, Doe said. When later removed from the orphanage four of every five of the children were unable to engage socially, according to research that followed the children into adulthood.
“If we can understand that mechanism of the closing of this critical developmental period, we could possibly reopen plasticity in children that were neglected or in adults who want to learn a new language or learn a new task,” Doe said.
That therapeutic potential is a long way off, the UO researchers said, but it is a major future goal. Any such drug that may be developed will require precise titration to find “the sweet spot for plasticity,” Ackerman said.
Her research will next move into similar studies in vertebrates, specifically in zebrafish, which were developed into a model research organism at the UO in the 1970s.
Co-authors with Ackerman and Doe were former UO undergraduate student Nelson A. Perez-Catalan, now in a postbaccalaureate program at the University of Chicago, and Marc R. Freeman, director and senior scientist of the Vollum Institute at Oregon Health and Science University in Portland.
Ackerman’s research involved mentoring UO undergraduate students, Doe noted. Perez-Catalan won a 2020 undergraduate research award for his work, which was detailed in his thesis “Jack-of-all-trades, The Role of Astrocytes in Circuit Formation and Plasticity.”
The Howard Hughes Medical Institute and National Institutes of Health funded the research through grants to Doe and Freeman, a former postdoctoral researcher in Doe’s UO lab. Ackerman was supported by a Milton Safenowitz Postdoctoral Fellowship awarded in 2017 by the ALS Association for research related to which amyotrophic lateral sclerosis, also known as Lou Gehrig’s Disease.
—By Jim Barlow, University Communications